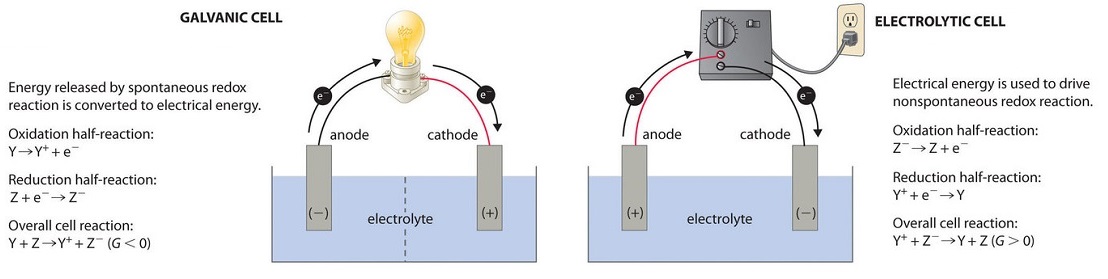
The Electrode Where Reduction Occurs Is Called the Anode
In an electrochemical cell the electrode that is the site of reduction is called the anode. The identity of the cathode and anode can be remembered by recognizing that positive ions or cations flow toward the cathode while negative ions or anions flow toward the anode.
17 1 Electrochemical Cells Chemistry Libretexts
Oxidation takes place at the right electrode so the right one is the anode.
. Part A The electrode where reduction occurs is called the anode. 100 20 ratings False. An anode is an electrode through which the conventional current enters into a polarized electrical device.
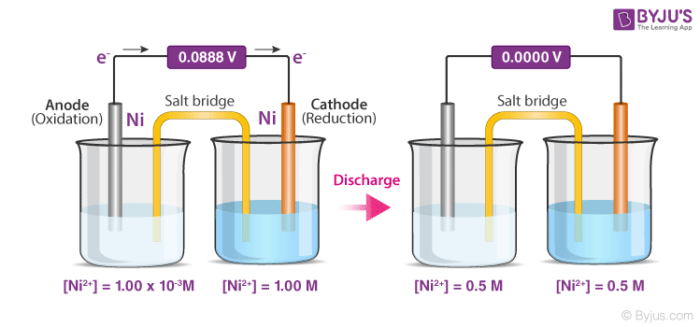
Battery is an electrochemical device that stores chemical energy for latter release as electricity in the form of direct electric current at a constant voltage. The electrode at which _____ occurs is called the cathode and the electrode where the _____ occurs is called the anode Oxidation neutralization Reduction neutralization Reduction oxidation Oxidation reduction oxidation neutralization reduction neutralization. Anode Cathode Voltage Reading so Anode Voltage Reading - Cathode 1a Find the Reduction Potentials for your metal ions to the metal.
The direction of conventional current in a circuit is opposite to the direction of electron flow so electrons flow. The electrode at which oxidation takes place is known as the anode while the electrode at which reduction take place is called the cathode. Fe3 to Zn2 0352 V Cu2 to Zn2 0270 V Co2 to Zn2 0254 V Al3 to Zn2 0479 V.
At the negative electrode cathode the reverse occurs. The identity of the cathode and anode can be remembered by recognizing that positive ions or cations flow toward the cathode while negative ions or anions flow toward the anode. The electrode at which oxidation takes place in a electrochemical cell is called the anode.
In an electrolytic cell oxidation occurs at the positive electrode also called the anode. 1b Calculate your Anode from your Measured Voltage Reading and the Reduction Potential voltage of each metal. The electrode at which reduction occurs is called the cathode.
Previous question Next question. View the full answer. O True O False Submit Request Answer.
By definition the anode in a galvanic cell is the electrode at which oxidation occurs and the cathode is the electrode at which reduction occurs. The electrode at which reduction occurs is called the cathode. This contrasts with a cathode an electrode through which conventional current leaves an electrical device.
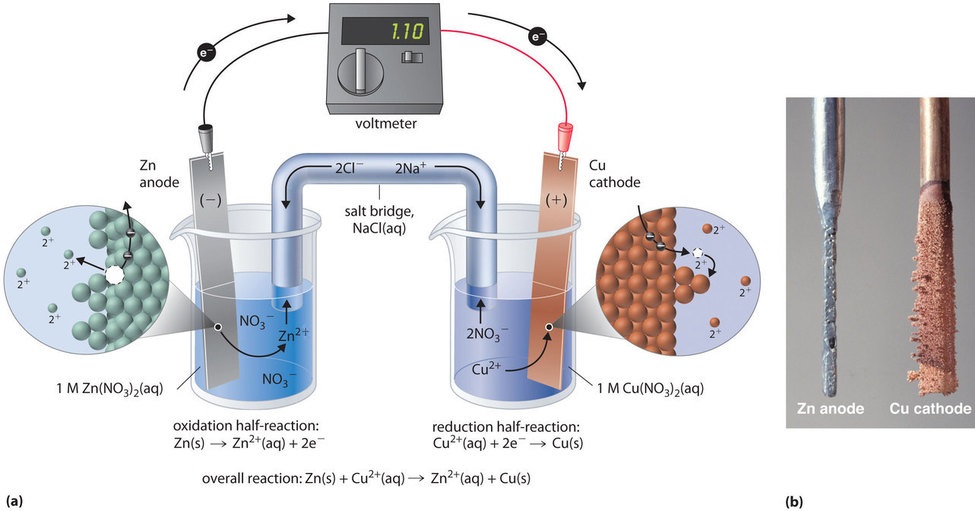
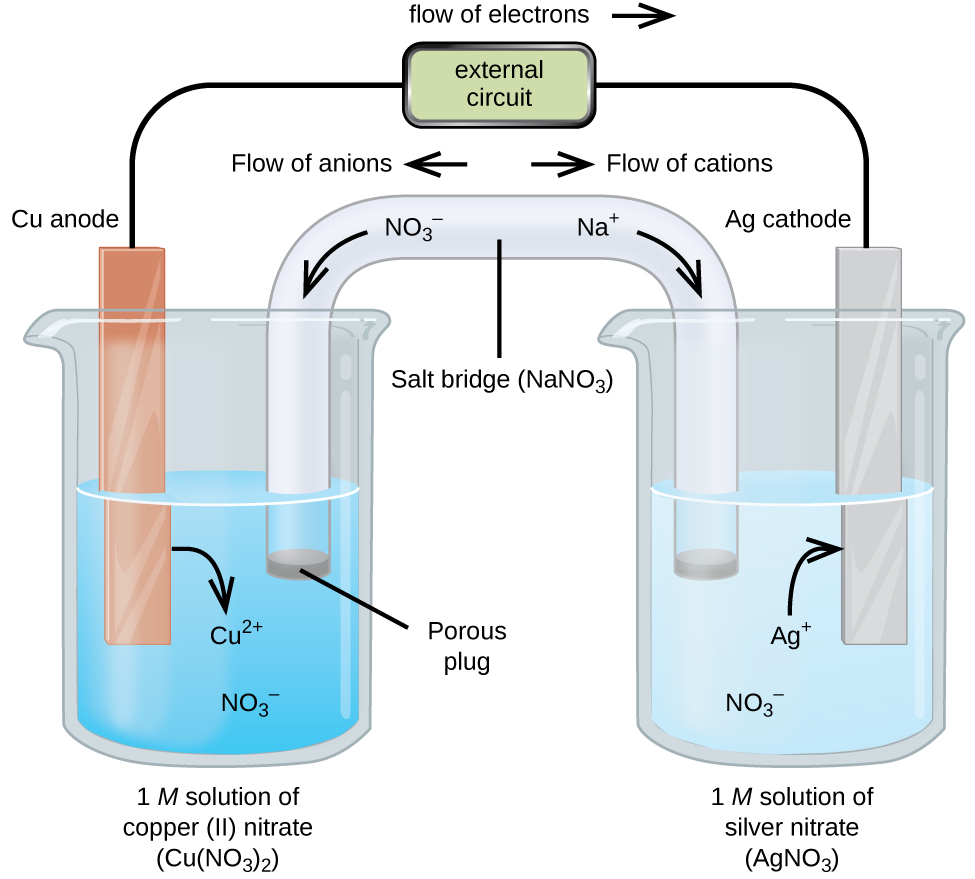
To complete the electrical circuit the solutions must be connected by a conducting medium through which the cations and anions can move from one electrode compartment to the other. The electrode where oxidation occurs is called anode while the electrode where reduction occurs is called cathode. The electrode at which oxidation takes place in a electrochemical cell is called the anode.
If you see galvanic cell reduction take place at the left electrode so the left one is the cathode. A common mnemonic is ACID for anode current into device.

Voltaic Cells Galvanic Cells Electrochemical Cells Chemtalk

Why Does Connecting A Battery S Positive Terminal To The Negative Terminal Of Another Battery Not Physical Chemistry Chemistry Lessons Environmental Chemistry
17 1 Electrochemical Cells Chemistry Libretexts
What Is The Type Of Reaction Occurring At Each Electrode Quora

Electrochemistry Charge Q A Property Of Matter Which Causes It To Experience The Electromagnetic Force Coulomb C The Quantity Of Charge Equal Ppt Download

Electrochemical Cells Chemistry Master

Anode Vs Cathode What S The Difference Physics Notes Galvanic Cell School Notes

Why Does Connecting A Battery S Positive Terminal To The Negative Terminal Of Another Battery Not Physical Chemistry Chemistry Lessons Environmental Chemistry

Oxidation Reduction Chemical Cells 4 Redox Reactions Oxidation Chemical

Learn About Electrochemistry Chegg Com

Click To Download Redox Rules Posters For Vce Chemistry Teaching Chemistry High School Science Ap Chemistry

Cathode And Anode Definition Examples And Key Differences

Click To Download Redox Rules Posters For Vce Chemistry Teaching Chemistry High School Science Ap Chemistry

Galvanic Cell Animation Zinc Silver Chemistry Lessons Teaching Chemistry Chemistry Experiments

Electrochemistry Flashcards Quizlet


Comments
Post a Comment